I marble chips are made from calcium carbonate hydrochloric acid calcium carbonate calcium chloride water carbon dioxideii the rate of reaction can be increased by increasing the concentration of hydrochloric acid which is the limiting factor in this equation.
Marble reacting with hydrochloric acid equation.
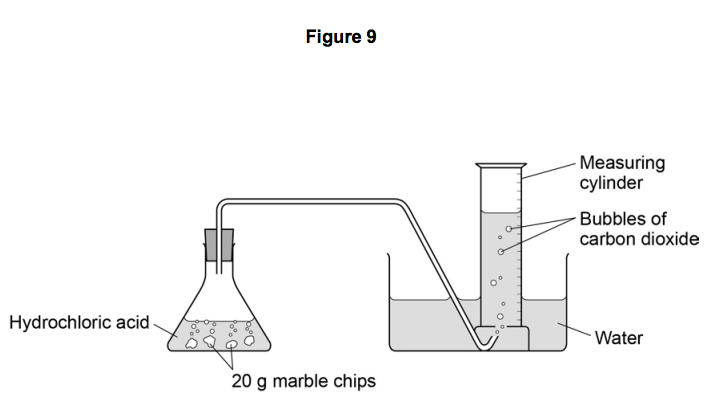
Hydrochloric acid 20ml 0 5m 1m 2m marble chips 2g per test large measuring cylinder plastic bowl 3 4 full of water rubber tubing glass conical flask stopwatch method.
Calcium carbonate is dissolved by hydrochloric acid thereby forming gaseous carbon dioxide.
Caco3 2hcl cacl2 h2co3.
The combined reactants have a higher chemical potential than the combined products i e.
Drop a small amount of dilute hydrochloric acid on an area of your sample that has been scratched by a nail.
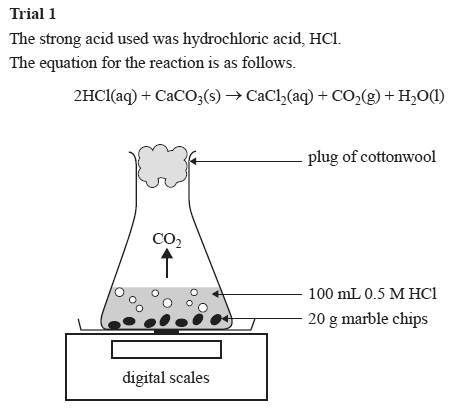
In words calcium carbonate reacts with hydrochloric acid to form calcium chloride water and carbon dioxide.
Hydrochloric acid is hcl.
Pieces of marble are thrown into hydro chloric acid.
Acids carbonates salts carbon dioxide water marble is caco3 2hcl caco3 cacl2 co2 h2o.
Marble chips are mostly made up of calcium carbonate which is a alkaline compound.
Caco3 2hcl cacl2 h2o co2.
When sulfurous sulfuric and nitric acids in polluted air and rain react with the calcite in marble and limestone the calcite dissolves.
Acid rain is one of the top degradation agents for marble artefacts around the world.
Powdered marble reacts with hydrochloric acid to release bubbles of carbon dioxide gas.
In exposed areas of buildings and statues we see roughened surfaces removal of material and loss of carved details.
Acid rain contains carbonic acid nitric acid and sulphuric acid co2 no2 and so2.
The reaction takes place spontaneously.
H2co3 decomposes easily into h2o and co2 so the equation is.
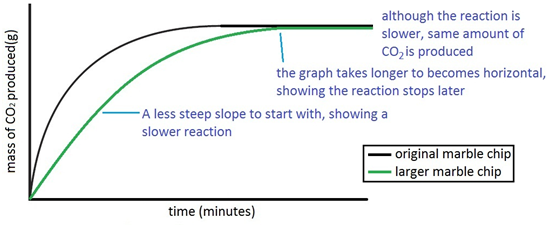
This experiment is to show how much carbon dioxide is produced during the reaction between an acid hydrochloric acid and marble.
Marble is especially sensitive to the degrading by acidic chemicals also to weathering.
Stone surface material may be lost all over or only in spots that are more reactive.
Marble is mostly made up of calcium carbonate which is caco3.
Hydrochloric acid to react with the marble chips independent variable marble chips to react with the acid dependent variable stopwatch to accurately time the experiment spatula to handle the marble chips measuring cylinder to precisely measure out different concentrations of hydryochloric acid electric balance to measure the mass g of the marble chips bung.
Marble reaction with hydrochloric acid.